-
Die Universität
- Herzlich willkommen
- Das sind wir
- Medien & PR
-
Studium
- Allgemein
- Studienangebot
- Campusleben
-
Forschung
- Profil
- Infrastruktur
- Kooperationen
- Services
-
Karriere
- Arbeitgeberin Med Uni Graz
- Potenziale
- Arbeitsumfeld
- Offene Stellen
-
Diagnostik
- Patient*innen
- Zuweiser*innen
- Gesundheitsthemen
- Gesundheitsinfrastruktur
Case of the Month
May 2026
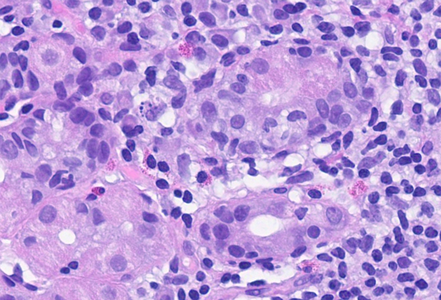
Gastric biopsies in a 43-year-old female, following Helicobacter pylori eradication.
Diagnosis
Early autoimmune gastritis.
Comment
A 43-year-old woman with history of Helicobacter pylori gastritis underwent gastric biopsies following eradication therapy 6 months before.
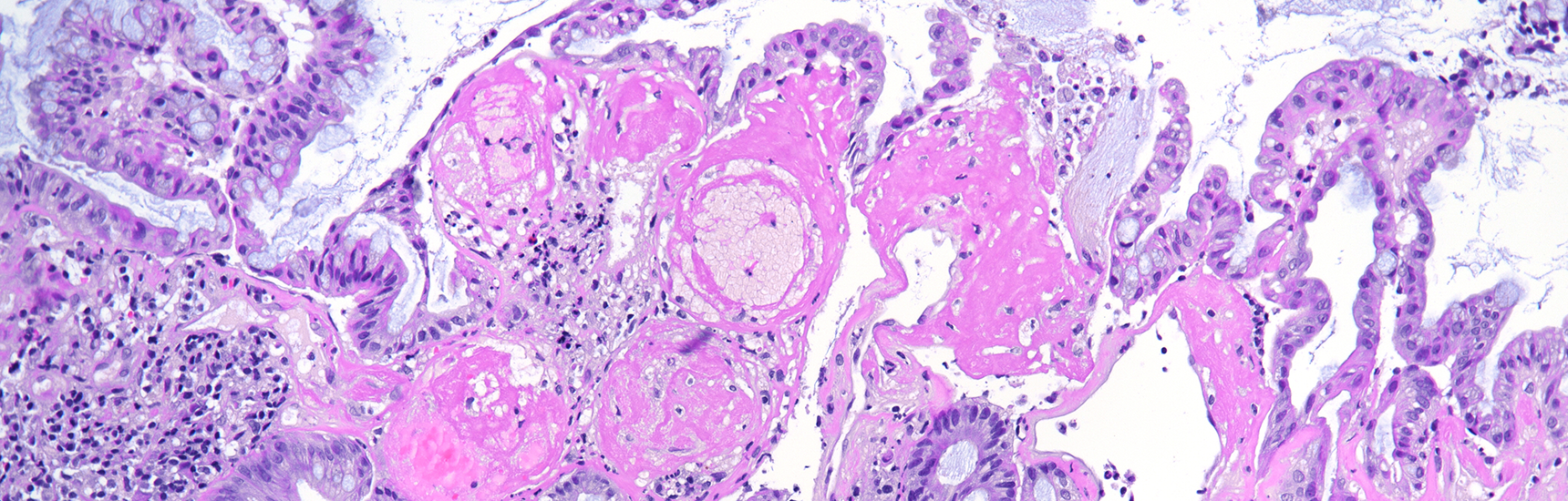
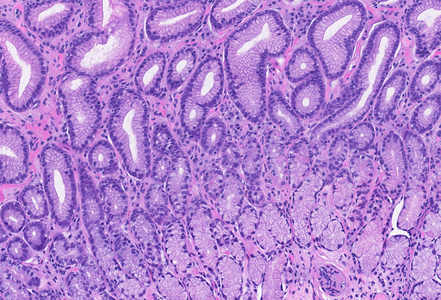
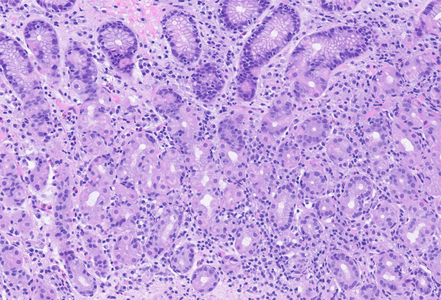
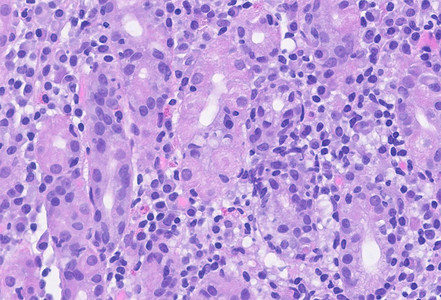
On histological evaluation of the gastric mucosa, mild inactive chronic antral gastritis was observed (Panel A), with no evidence of Helicobacter pylori on immunohistochemistry, thus qualifying for “post-HP gastritis”. Chronic inflammation was more pronounced in the gastric corpus, with a lymphoplasmacytic infiltrate in the lower lamina propria, within the glandular component (Panel B), a focal increase in eosinophil count and intraepithelial lymphocytes (“emperipolesis”), with parietal cell destruction and apoptosis (Panels C and D). Although these histological findings do not represent the full/end-stage spectrum of the disorder, they represent the characteristic morphology of early-stage autoimmune gastritis.
Autoimmune gastritis is an inflammatory disorder that usually develops independent of prior Helicobacter pylori infection and shows female predominance, younger age and association with other autoimmune diseases. Autoimmune gastritis may, however, also develop as a consequence of Helicobacter pylori infection, in older age and with more or less equal gender distribution. In these cases, a molecular mimicry between antigens of Helicobacter pylori and the proton pump (H⁺/K⁺-ATPase) of parietal cells has been suggested.
Morphologically, autoimmune gastritis is characterized by CD4⁺ T-cell mediated injury to the parietal cells, ultimately leading to their loss and to an “antrum-like” transformation of oxyntic mucosa (“pseudopyloric metaplasia”). The consequent hypochlorhydria and compensatory antral G-cell hyperplasia, in turn, promote proliferation of enterochromaffin-like cells, which can be identified on chromogranin A immunostaining by linear chains (typically >6 cells) or nodular aggregates. Because the parietal cells are only present in the oxyntic mucosa, the typical basal inflammation and atrophy, with intestinal, pseudopyloric and pancreatic metaplasia is confined to the gastric corpus and fundus, with the antrum showing little to no alterations. In our case, the mild antral post-HP gastritis may indicate Helicobacter pylori dependent disease development.
While fully developed autoimmune gastritis exhibits these features clearly, the early stages of the disease can be easily overlooked, particularly in the setting of prior Helicobacter pylori infection, where concomitant antral and corpus inflammation may be misinterpreted as entirely infection related.
For further reading
Rugge M, Genta RM, Malfertheiner P, et al. RE.GA.IN.: the Real-world Gastritis Initiative–updating the updates. Gut. 2024 Feb 23;73(3):407–441.
Bettington M, Brown I. Autoimmune gastritis: novel clues to histological diagnosis. Pathology. 2013 Feb;45(2):145–149.
Pittman ME, Voltaggio L, Bhaijee F, et al. Autoimmune metaplastic atrophic gastritis: recognizing precursor lesions for appropriate patient evaluation. Am J Surg Pathol. 2015 Dec;39(12):1671–1681.
Presented by
Dr. Ana Lucia Alves, Coimbra, Portugal, and Dr. Cord Langner, Graz, Austria.